AMPED EFRC
The Alliance for Molecular PhotoElectrode Design for Solar Fuels is an Energy Frontier Research Center headquartered in the Chemistry Department at the University of North Carolina at Chapel Hill.
The Center includes research partners at the University of North Carolina at Chapel Hill, the University of Texas at San Antonio, and Georgia Institute of Technology and has been funded since 2009 by the U.S. Department of Energy Office of Science, Office of Basic Energy Sciences
SCIENTIFIC MISSION
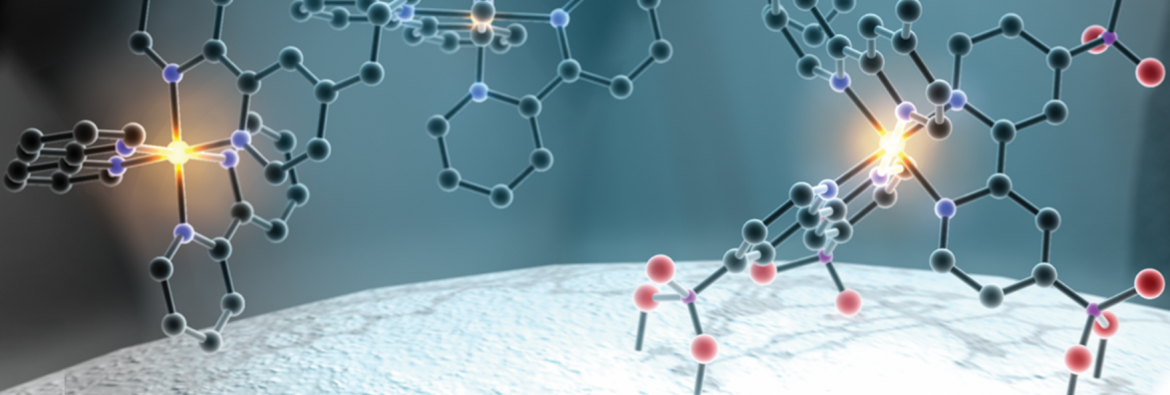
Sunlight is the one renewable source that could by itself meet all the energy demands of our growing world economy. For solar energy to reach its full potential it must be coupled to a storage method, which can be achieved by conversion of sunlight into chemical energy stored in bonds of molecules called solar fuels.
Inspired by natural photosynthesis, the mission of the AMPED EFRC is to develop the fundamental molecular basis for solar-driven water oxidation and carbon dioxide reduction catalysis.
|
|
|